Simanaitis Says
On cars, old, new and future; science & technology; vintage airplanes, computer flight simulation of them; Sherlockiana; our English language; travel; and other stuff
SOAP BUBBLES AND WINE LEGS
SOAP BUBBLES make for a fascinating physics lab, even on your own table top. Knvul Sheikh gives proof of this in “Watch Soap Bubbles Turn Into Tiny SnowGlobes as They Freeze,” The New York Times, June 18, 2019.
Plateau’s Achievement. Indeed, this is the third appearance of soap bubbles here at SimanaitisSays. Back in the middle 1800s, the French physicist Joseph Plateau studied how soap bubbles interact: If they’re the same volume, their intersection is a circular disc.

Image from the University of California Berkeley
Plateau figured out their intersection even when the two bubbles differ in size. His geometric construction is all the more elegant, given that Plateau was blind at the time. Back when I was teaching, I used this as counterexample to kids saying “I can’t do that problem because I can’t visualize it.”
Karen Uhlenbeck’s Abel Prize. Soap bubbles were part of the research that garnered American mathematician Karen Uhlenbeck the 2019 Abel Prize; equivalent, in a sense, to a mathematics Nobel Prize.

Image by Kym Cox/Science Source from The New York Times, April 8, 2019.
Uhlenbeck’s work in geometric partial differential equations analyzed, among other things, how soap films behaved in higher-dimensional curved space. (Now there’s something that takes visualizing.)
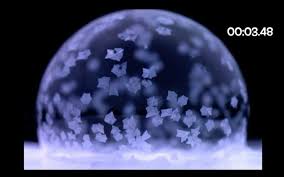
Freezing Bubbles. The freezing of bubbles is an example of heat transfer, how matter behaves when its temperature changes. As described by Knvul Sheikh, “… when soap bubbles freeze, lacy crystals break off from cooler points on the surface and swirl around on currents of warmer liquid.” As Virginia Tech’s Jonathan Boreyko said to Sheikh, “We’ve seen the unique freezing dynamics of bubbles in nature, but we’ve never understood the physics behind it.”
Boreyko and his colleagues, S. Farzad Ahmadi, Saurabh Nath, Christian M. Kinglett, and Pengtao Yue, published their findings, “How Soap Bubbles Freeze,” in Nature Communications 10, Article 2531, 2019.
The researchers note, “Droplets or puddles tend to freeze from the propagation of a single freeze front. In contrast, videographers have shown that as soap bubbles freeze, a plethora of growing ice crystals can swirl around in a beautiful effect visually reminiscent of a snow globe.”
Sheikh writes, “The team started by pipetting tiny soap bubbles onto an ice block that they chilled to minus 4 Fahrenheit and kept in a walk-in freezer. Using high-speed cameras, the team then filmed the soap bubbles as they froze from the bottom up.”
A Quick Show. At first, the boundary between frozen and unfrozen parts, the “freeze front,” inched up the surface. “But,” Sheikh says, “within milliseconds, chunks of ice crystals started breaking off from the freeze front. They were swept up by tiny water streams, known as Marangoni currents, until hundreds of ice crystals danced across the bubble.”

Bubbles freezing. Images by John Lloyd from The New York Times, June 18, 2019. Sheikh’s article also contains a compelling video.
“After about 10 seconds,” Sheikh writes, “the Marangoni flow dissipated and died out as the floating ice grew and crystals became interlocked. At that point, the rest of the bubble froze over.”
I was curious about Marangoni and learned more from several sources. For instance, Marangoni currents explain why wine appears to leave “legs” behind in your glass as you drink.

Image from fyfluiddynamics.com.
Wine Traveler explains that these traces, also known as “tears of wine,” are a product of alcohol, evaporation, water, and surface tension. In particular, they have nothing to do with how sediment-packed the wine is.
An onophile friend used to quip, “This wine has good legs…. and a nice smile too.”
More on Marangoni. To give the phenomenon its full name, the Gibbs-Marangoni Effect honors Italian physicist Carlo Marangoni and American scientist Joshua Willard Gibbs. Marangoni discussed surface-tension phenomena in his 1865 doctoral thesis; Gibbs gave it a full mathematical treatment in his On the Equilibrium of Heterogeneous Substances. 1875–1878.
The images are beautiful, the physics and mathematics are elegant. And there’s practicality as well. Sheikh notes, “The study could also have applications for flash freezing food, creating tastier ice cream, or even developing antifreeze materials.”
I’m all for tastier ice cream, though I believe this bar is already quite high. I love Mexican Chocolate. ds
© Dennis Simanaitis, SimanaitisSays, 2019


Besides tastier ice cream, don’t forget wines with smiles, or wines that make you smile.