Simanaitis Says
On cars, old, new and future; science & technology; vintage airplanes, computer flight simulation of them; Sherlockiana; our English language; travel; and other stuff
MASSIVE DIAMONDS, PART 1
A MASSIVE DIAMOND of 404.2 carats (almost 1/5 lb.) made the December 16, 2016, cover of Science, the weekly magazine of the American Association for the Advancement of Science. It’s quite the rock, and Science gives newly discovered details of its origin. These get technical pretty quickly, which got me refreshing my memory bank about such precious gems. And, wouldn’t you know, I gleaned enough stuff to warrant two items here at SimanaitisSays.
Today, the Science aspects. Tomorrow, tidbits of massive diamonds, which, I admit, sounds oxymoronic.

This 404.2-carat rough diamond, 2 3/4 inches tall, was recovered from the LuLo Mine, Angola, in February 2016. This and other images from Science, both print and online.
The Science technical paper ”Large Gem Diamonds From Metallic Liquid in Earth’s Deep Mantle,” by Evan M. Smith et al, has an online abstract. Also, ”Earth’s Rarest Diamonds Formed in Pockets of Liquid Metal,” by Sid Perkins, offers an online overview of the research.
The word diamond comes from the Greek, ἀδάμας – adámas, “unbreakable.” However, diamonds aren’t exactly “forever.” Chemically, they’re a metastable form of carbon, the metastable moniker implying that their “diamond lattice” crystal structure is less stable than graphite’s. Rest easy, though, because in ordinary conditions the conversion from diamond to graphite is negligibly slow.
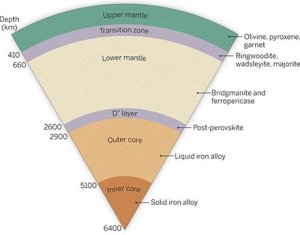
Natural diamonds are formed in the Earth’s mantle at extremes of heat and pressure. The mantle is immediately beneath the Earth’s paper-thin outmost layer of about 3 to 30 miles down. And, indeed, “paper-thin” is a good analogy: On an eight-inch globe, the crust would have the thickness of a sheet of paper.
The mantle, about 1800 miles thick, makes up almost 7/8 of the Earth’s volume. It’s semi-solid with metallic liquids under great pressure and high temperature. This latest research posits that certain diamonds, the Type II variety, arise in these metal-saturated regions of the mantle.
Type II diamonds are those with no measurable nitrogen impurities; they make up less than two percent of natural diamonds. They’re typically large, relatively pure and irregularly shaped.
Evan Smith of the Gemological Institute of America and his colleagues from the U.S., Italy and South Africa examined 53 Type II diamonds. For 38 of these diamonds, the only inclusions were of a magnetic, metallic character. One conjecture of this finding is that such diamonds form within the mantle at depths of 255 to 570 miles. Another suggests how these massive gems are formed.
The researchers say that in the mantle a liquid metal composition evolves of Iron, Nickel, Carbon and Sulfur. The carbon crystalizes within these metallic liquid pockets. These diamonds then physically separate from their growth environment and work their way to the crust through volcanic action.
This complex path leads from the Earth’s mantle to what Science magazine calls “A Geologist’s Best Friend.”
Tomorrow, I describe the goings-on of several of these massive diamonds. Involved, in one manner or another, are various European royalty, a thief or two, F. Scott Fitzgerald and Mickey Mouse. ds
© Dennis Simanaitis, SimanaitisSays.com, 2017
Related
Information
This entry was posted on January 1, 2017 by simanaitissays in Sci-Tech and tagged "Earth's Rarest Diamonds Formed in Pockets of Liquid Metal" Sid Perkins AAAS Science, "Large Gem Diamonds from Metallic Liquids in Earth's Deep Mantle" Evan M. Smith et al, AAAS Science.Shortlink
https://wp.me/p2ETap-5oqCategories
Recent Posts
Archives
- May 2026
- April 2026
- March 2026
- February 2026
- January 2026
- December 2025
- November 2025
- October 2025
- September 2025
- August 2025
- July 2025
- June 2025
- May 2025
- April 2025
- March 2025
- February 2025
- January 2025
- December 2024
- November 2024
- October 2024
- September 2024
- August 2024
- July 2024
- June 2024
- May 2024
- April 2024
- March 2024
- February 2024
- January 2024
- December 2023
- November 2023
- October 2023
- September 2023
- August 2023
- July 2023
- June 2023
- May 2023
- April 2023
- March 2023
- February 2023
- January 2023
- December 2022
- November 2022
- October 2022
- September 2022
- August 2022
- July 2022
- June 2022
- May 2022
- April 2022
- March 2022
- February 2022
- January 2022
- December 2021
- November 2021
- October 2021
- September 2021
- August 2021
- July 2021
- June 2021
- May 2021
- April 2021
- March 2021
- February 2021
- January 2021
- December 2020
- November 2020
- October 2020
- September 2020
- August 2020
- July 2020
- June 2020
- May 2020
- April 2020
- March 2020
- February 2020
- January 2020
- December 2019
- November 2019
- October 2019
- September 2019
- August 2019
- July 2019
- June 2019
- May 2019
- April 2019
- March 2019
- February 2019
- January 2019
- December 2018
- November 2018
- October 2018
- September 2018
- August 2018
- July 2018
- June 2018
- May 2018
- April 2018
- March 2018
- February 2018
- January 2018
- December 2017
- November 2017
- October 2017
- September 2017
- August 2017
- July 2017
- June 2017
- May 2017
- April 2017
- March 2017
- February 2017
- January 2017
- December 2016
- November 2016
- October 2016
- September 2016
- August 2016
- July 2016
- June 2016
- May 2016
- April 2016
- March 2016
- February 2016
- January 2016
- December 2015
- November 2015
- October 2015
- September 2015
- August 2015
- July 2015
- June 2015
- May 2015
- April 2015
- March 2015
- February 2015
- January 2015
- December 2014
- November 2014
- October 2014
- September 2014
- August 2014
- July 2014
- June 2014
- May 2014
- April 2014
- March 2014
- February 2014
- January 2014
- December 2013
- November 2013
- October 2013
- September 2013
- August 2013
- July 2013
- June 2013
- May 2013
- April 2013
- March 2013
- February 2013
- January 2013
- December 2012
- November 2012
- October 2012
- September 2012
- August 2012