Simanaitis Says
On cars, old, new and future; science & technology; vintage airplanes, computer flight simulation of them; Sherlockiana; our English language; travel; and other stuff
H2 PRODUCTION
THE WORD “breakthrough” can get overworked in science news, but Virginia Tech researchers may have a legitimate claim to it. They’ve discovered an efficient way to extract significant quantities of hydrogen from any plant material. Noted one of the researchers, “Hydrogen is one of the most important biofuels of the future.”
See http://goo.gl/56M6J for the full Virginia Tech release. Here, I’ll summarize it together with some of my own amplification.
As is often noted, hydrogen is the most abundant element on Earth, indeed, the most abundant in the universe. What a perfect choice for an energy carrier. Except for one attribute: Hydrogen is also one of the most promiscuous elements. It likes to couple with just about anything, thus complicating its separation and production.
In fact, a hydrogen fuel cell depends on this promiscuity. Hydrogen has a thing for oxygen in the air. Set up the two through the right membranes in the presence of the right catalyst, and the products are water (H2O), a little heat—and electricity. Quite enough electricity to power a fuel-cell electric vehicle. That is, an FCEV is an EV carrying hydrogen to make its own E.

FCEV manufacturers in the 2015-2017 timeframe. Clockwise from the top left, Honda Clarity, Hyundai i-Blue concept, Mercedes-Benz F-Cell, Nissan Terra Concept FCEV and Toyota FCV.
Automakers are introducing FCEVs in 2015-2017. A refueling infrastructure is evolving. But whence the hydrogen?
There’s no shortage of the stuff today, but it’s typically a byproduct of natural gas production through an energy-intensive process that releases considerable amounts of CO2. Hydrogen-highway skeptics, including former Energy Secretary Steven Chu, have questioned this less-than-ideal sourcing of a potential transportation fuel.
Enter the Virginia Tech researchers. In theory, they knew that a common plant sugar, xylose, exposed to certain enzymes could produce a large quantity of hydrogen. After seven years work, these researchers transformed theory to reality.

Associate Professor Y.H. Percival Zhang is principal researcher in this work at Virginia Tech’s College of Agricultural and Life Sciences and College of Engineering.
The environmentally friendly process uses renewable—and, an important point, non-food—sources as input. What’s more, it requires no costly metallic catalysts. Its biocatalysts are a group of enzymes not occurring naturally. However, researchers figured out how to isolate them from microorganisms, some of which grow at around the boiling point of water.
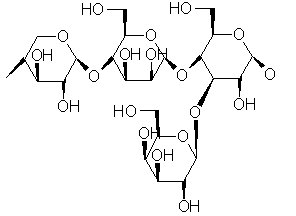
Hemicellulose is present in almost all plant cell walls. The trick to hydrogen production is freeing up those H’s.
The word “xylose” is from the Greek, ξυλος, xylos, meaning “wood.” Xylose is a precursor of xylan, a hemicellulose that makes up as much as 30 percent of plant cell walls. One source of xylose is agricultural waste, plant stalks and the like. The innovative enzyme blend liberates high-purity hydrogen from this xylose at a rate three times that of previous processes. Conditions are mild, 122 degrees Celsius and normal atmospheric pressure.
Today’s hydrogen is used in refining petroleum products as well as in manufacturing ammonia for fertilizers. It’s already an annual $100 billion market, with potential growth coming from an evolving use in transportation. It’s said the Virginia Tech process could find its way to the marketplace in as little as three years.
Perfect timing for those FCEVs from Honda, Hyundai, Mercedes-Benz, Nissan and Toyota. ds
© Dennis Simanaitis, SimanaitisSays.com, 2013