Simanaitis Says
On cars, old, new and future; science & technology; vintage airplanes, computer flight simulation of them; Sherlockiana; our English language; travel; and other stuff
WHAT DO MUSSELS HAVE IN COMMON WITH MODEL T FORDS?
OR, IF YOU PREFER modern technology, mussels and jet engine turbines? The answer is the metal vanadium. Here are tidbits on this commonality gleaned from Science magazine and from my usual Internet sleuthing.

Mussels. Johnathan J. Wilker reports in “Rare Metal, Precious Adhesion,” Science magazine, October 8, 2021, that “marine mussels use vanadium, along with iron, to construct their protein-based adhesive.”

Model T Fords. As described by Lindsay Brooke in Popular Mechanics, September 25, 2000, “This lightweight, durable, easily machined steel alloy was developed in Europe and used on racing cars and premium-priced vehicles before Ford became aware of it around 1905. He recognized that the material’s high tensile strength (nearly three times greater than cheaper, lower-grade steels) would allow him to make a stronger, lighter, better performing car.”

Brooke continued, “Ford and his small team of engineers first tested vanadium steel in their Model N and S cars during 1906-1907, before deciding to use it in many of the Model T’s critical highly stressed parts including the crankshaft, forged front axle, and wheel spindles.”
Today’s Technology. In October 2017, Rick Mills described vanadium as “The Metal We Can’t Do Without and Don’t Produce.” He noted, “Just two pounds of vanadium added to a tonne of steel doubles its strength, so it is unsurprising that 80% of vanadium is used to make ferrovanadium—a steel additive…. Twenty years ago no vanadium went into cars, versus around 45 percent today.”
“By 2025,” Mills predicted back in 2017, “it’s estimated that 85 percent of all automobiles will incorporate vanadium alloy to reduce their weight, thereby increasing their fuel efficiency to conform to stringent fuel economy standards set by the US EPA. Who would have thought any material could make steel ‘greener’?”
What’s more, a thin layer of vanadium bonds titanium to steel, making it ideal for aerospace applications. Mixing titanium with vanadium and iron strengthens and adds durability to turbines that spin up to 70,000 rpm.
Today’s Resource Challenge. Wikipedia describes the challenge: Vanadium is abundant, yet rare. It’s detected spectroscopically in the Sun and sometimes in other stars; in the universe it’s nearly as common as copper or zinc.
It’s the 20th most abundant element in the Earth’s crust. However, it’s typically combined with other elements in some 65 different minerals. Crucially, more than 97 percent of the world’s vanadium comes from only three countries: South Africa, north-western China, and eastern Russia.
Yet, Wikipedia also notes, “The vanadyl ion is abundant in seawater….” Which brings us to mussels.
Mussels’ Two-Tube Adhesive. Mussels have evolved an approach for manufacturing an iron-vanadium adhesive comparable to two-tube epoxy glues. Details are given in “Microfluidic-like Fabrication of Metal Ion-cured Bioadhesives by Mussels,” Tobias Priemel, et al, Science, October 8, 2021.
From their Abstract: “To anchor in seashore habitats, mussels fabricate adhesive byssus fibers that are mechanically reinforced by protein-metal coordination mediated by 3,4-dihydroxyphenylalanine (DOPA). The mechanism by which metal ions are integrated during byssus formation remains unknown.”
“In this study,” they continue, “we investigated the byssus formation process in the blue mussel, Mytilus edulis, combining traditional and advanced methods to identify how and when metals are incorporated. Mussels store iron and vanadium ions in intracellular metal storage particles (MSPs) complexed with previously unknown catechol-based biomolecules.”
This, it’s noted, is akin to our two-tube epoxies mixed immediately upon application.
“During adhesive formation,” the researchers explain, “stockpiled secretory vesicles containing concentrated fluid proteins are mixed with MSPs within a microfluidic-like network of interconnected channels where they coalesce, forming protein-metal bonds within the nascent byssus.”
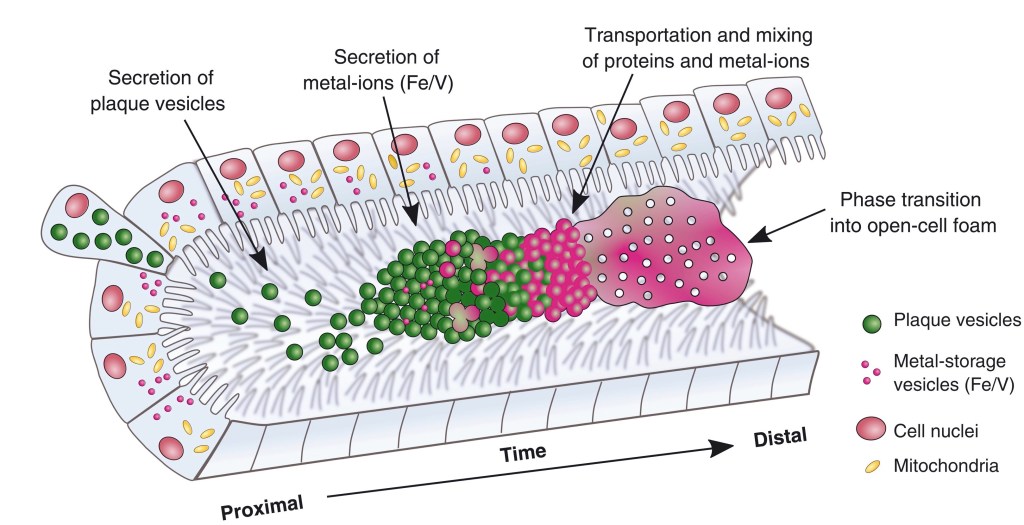
They conclude, “These findings advance our understanding of metal use in biological materials with implications for next-generation metallopolymers and adhesives.”
All this from mussels sticking together in the intertidal. ds
© Dennis Simanaitis, SimanaitisSays.com, 2021
Related
Information
This entry was posted on October 27, 2021 by simanaitissays in Sci-Tech and tagged "Rare Metal Precious Adhesion" Johnathan J. Wilker "Science", Microfluidic-like Fabrication of Metal Ion-cured Bioadhesives by Mussels" Priemel et al "Science", Model T Ford vanadium-steel components, mussels make their own two-tube adhesive with iron and vanadium, vanadium-steel three times strength of steel.Shortlink
https://wp.me/p2ETap-d23Categories
Recent Posts
Archives
- May 2026
- April 2026
- March 2026
- February 2026
- January 2026
- December 2025
- November 2025
- October 2025
- September 2025
- August 2025
- July 2025
- June 2025
- May 2025
- April 2025
- March 2025
- February 2025
- January 2025
- December 2024
- November 2024
- October 2024
- September 2024
- August 2024
- July 2024
- June 2024
- May 2024
- April 2024
- March 2024
- February 2024
- January 2024
- December 2023
- November 2023
- October 2023
- September 2023
- August 2023
- July 2023
- June 2023
- May 2023
- April 2023
- March 2023
- February 2023
- January 2023
- December 2022
- November 2022
- October 2022
- September 2022
- August 2022
- July 2022
- June 2022
- May 2022
- April 2022
- March 2022
- February 2022
- January 2022
- December 2021
- November 2021
- October 2021
- September 2021
- August 2021
- July 2021
- June 2021
- May 2021
- April 2021
- March 2021
- February 2021
- January 2021
- December 2020
- November 2020
- October 2020
- September 2020
- August 2020
- July 2020
- June 2020
- May 2020
- April 2020
- March 2020
- February 2020
- January 2020
- December 2019
- November 2019
- October 2019
- September 2019
- August 2019
- July 2019
- June 2019
- May 2019
- April 2019
- March 2019
- February 2019
- January 2019
- December 2018
- November 2018
- October 2018
- September 2018
- August 2018
- July 2018
- June 2018
- May 2018
- April 2018
- March 2018
- February 2018
- January 2018
- December 2017
- November 2017
- October 2017
- September 2017
- August 2017
- July 2017
- June 2017
- May 2017
- April 2017
- March 2017
- February 2017
- January 2017
- December 2016
- November 2016
- October 2016
- September 2016
- August 2016
- July 2016
- June 2016
- May 2016
- April 2016
- March 2016
- February 2016
- January 2016
- December 2015
- November 2015
- October 2015
- September 2015
- August 2015
- July 2015
- June 2015
- May 2015
- April 2015
- March 2015
- February 2015
- January 2015
- December 2014
- November 2014
- October 2014
- September 2014
- August 2014
- July 2014
- June 2014
- May 2014
- April 2014
- March 2014
- February 2014
- January 2014
- December 2013
- November 2013
- October 2013
- September 2013
- August 2013
- July 2013
- June 2013
- May 2013
- April 2013
- March 2013
- February 2013
- January 2013
- December 2012
- November 2012
- October 2012
- September 2012
- August 2012
