Simanaitis Says
On cars, old, new and future; science & technology; vintage airplanes, computer flight simulation of them; Sherlockiana; our English language; travel; and other stuff
LITHIUM-ION BATTERIES
NO PUN intended, but we need to go a little deeper with the grounding of Boeing 787 Dreamliners, their fire and smoke emergencies having been attributed to the aircraft’s use of lithium-ion batteries. Some commentators blame “the same technology used in cars,” and then they cite fires with the Fisker Karma and Chevrolet Volt.
It’s more complicated than this.
The confusion comes from the fact that there’s a variety of lithium battery chemistries, each with a different cathode material and resulting performance tradeoffs.
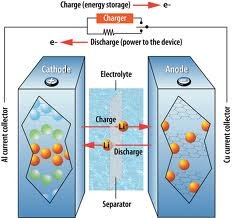
What do these batteries have in common? As with all batteries, a lithium-ion rechargeable battery works by shuttling electrical charge between two electrodes, a positively charged cathode and negatively charged anode.
Fully charged, the lithium ions hang out at the anode. Flip a switch, and (negative) electrons in the anode are attracted through this external circuit where they perform work before joining the (positively charged) cathode. This electron travel causes the lithium ions to migrate through an electrolyte to the cathode. The electricity continues to flow until migration ceases—i.e., until the battery needs recharging.
In recharging, an applied external voltage sends electrons back from cathode to anode; the lithium ions do their thing by returning to the anode and teaming up with the electrons again.
This same routine occurs in any lithium-ion battery. Also, the anode material is typically carbon. But lithium chemistries diverge in their choice of cathode material—with plenty of tradeoffs.
A serious tradeoff—indeed, a hazard—is thermal runaway, the bête noir of lithium batteries. Charging any battery causes heat and, with some chemistries, too much heat can lead to higher and higher temperatures, even to the point of explosion.
A classic lithium battery story: A parachute tester finds himself plummeting downward when he sees another guy coming up toward him. The parachuter yells, “Hey, do you know anything about parachutes??” “No,” yells the guy heading up, “do you know anything about lithium batteries??”
Thermal runaway is profoundly related to cathode choice. And Mother Nature, sometimes being a real mother, rewards the more efficient cathode materials with higher propensities of thermal runaway. This is countered by complex techniques involving circuitry, materials, recharge strategies and failure modes.
Lithium cobalt oxide, LiCoO2, is a very efficient cathode material, with risk of thermal runaway mitigated by appropriate engineering. It has been the cathode of choice for batteries in mobile phones and laptops—thus, giving an excellent data base of battery use. (Early laptop fires, for example, were traced to impurities causing shorts, not thermal runaway per se.)
For reasons of high efficiency, the Boeing 787 Dreamliner’s batteries are also of the cobalt oxide variety.
By contrast, the only automotive application of cobalt oxide chemistry was with the Tesla Roadster. The Chevrolet Volt’s cathodes are lithium manganese spinel. The Fisker Karma has another conservative cathode choice, lithium iron phosphate. These automotive cathode choices trade some efficiency for other benefits, including avoidance of anything resembling thermal runaway.
The Volt fire occurred three weeks after a side-impact crash test. It was traced to a ruptured battery coolant line. Also contributing was a shortcoming of test protocol for such vehicles (a conventional car’s leaking gasoline tank, for example, wouldn’t have been left for three weeks).
One Fisker Karma fire was attributed to a faulty cooling fan, unrelated to the car’s battery pack. Others involved 17 Karmas that were submerged in Hurricane Sandy waters. The fires were traced to salt water intrusion of electrical control modules. ds
© Dennis Simanaitis, SimanaitisSays.com, 2013




Excellent! This will be presented to our FAA Airframe & Powerplant Mechanic certification class on Tuesday (with the appropriate credits and an encouragement to subscribe to SS, of course).
Thanks You, Sir
Nice Dennis. The key, as you point out is the battery management system (BMS) that keeps everything under control (or perhaps doesn’t in the Boeing case…)
As always a thorough, non-hysterical analysis of a complex issue – at last for most media – that sheds light, not heat on a subject.
Hey Dennis, what do you think of Elon Musk’s comments about the 787 batteries?
In Tesla’s favor, I don’t know of any giant holes in Silicon Valley. As Kevin noted here, a battery management system is key.
Yet, a cobalt oxide electrode is inherently the most risky type. And Tesla’s latest models use more conservative technology.
Great explained article.
You comment here that “…Yet, a cobalt oxide electrode is inherently the most risky type. And Tesla’s latest models use more conservative technology…”
However, in the article quoting Elon Musk in FLightGlobal, they state “…Both Boeing and Tesla use batteries fueled by lithium cobalt oxide, which is among the most energy-dense and flammable chemistries of lithium-ion batteries on the market. While Boeing elected to use a battery with a grouping of eight large cells, Tesla’s batteries contain thousands of smaller cells that are independently separated to prevent fire in a single cell from harming the surrounding ones…”
article http://www.flightglobal.com/news/articles/elon-musk-boeing-787-battery-fundamentally-unsafe-381627/
So what is the more conservative technology that Tesla’s latest models use?
Musk is running an automotive Ponzi scheme. Beware.
Hi, Maramusa,
Thanks for your kind comments.
From what I can find, the Tesla Model S uses cathodes of nickel-cobalt-aluminum-oxide (NCA), apparently more conservative than the Roadster’s cobalt-oxide variety.
As Musk noted, Tesla continues to use a multiplicity (thousands) of small batteries, as opposed to fewer, larger ones. True, this mitigates one aspect of cascading problems. On the other hand, it complicates the BMS (Battery Management System) because there are thousands of cells to connect, monitor and control.
At a recent SAE Hybrid/EV meeting (see several mini-essays here on it), specialists spoke of BMS as being especially crucial in the Boeing matter.